 题目内容
(请给出正确答案)
题目内容
(请给出正确答案)
4 Ag (s) + 4 HCl(aq) + 4O2(g) ? 4 AgCl(s) + 2H2O(l),当c(HCl) = 6.0 mol/dm3, p(O2) = 100 kPa时,298 K下该反应的E和 ΔrGθM分别是()。
A.1.099 V、-388.6 kJ/mol
B. 1.053 V、388.6 kJ/mol
C. 1.190 V、-144.8 kJ/mol
D. 1.053 V、-388.6 kJ/mol
 题目内容
(请给出正确答案)
题目内容
(请给出正确答案)
A.1.099 V、-388.6 kJ/mol
B. 1.053 V、388.6 kJ/mol
C. 1.190 V、-144.8 kJ/mol
D. 1.053 V、-388.6 kJ/mol
 更多“4 Ag (s) + 4 HCl(aq) + 4O2(g) …”相关的问题
更多“4 Ag (s) + 4 HCl(aq) + 4O2(g) …”相关的问题
A.Ag, AgCl (s) | HCl(aq) | Cl2(g ,p),Pt
B.Pt, Cl2(g , p) | HCl(aq) |AgNO3 (aq) | Ag
C.Ag |AgNO3 (aq)| HCl(aq) | AgCl (s) , Ag
D.Ag, AgCl (s) | HCl(aq) | HCl(aq) | AgCl (s) , Ag
A.Ag|AgCl(s)|HCl(aq)|Cl2 (g,100kPa)|Pt
B.Pt| Cl2 (g,100kPa)| HCl(aq)||AgNO3 (aq)|Ag
C.Ag|AgNO3 (aq)| HCl(aq)|AgCl(s)|Ag
D.Ag|AgCl(s)| HCl(a1)||HCl(a2)|AgCl(s)Ag
A.Ag(s)|AgCl(s)|HCl(aq)|H2(P)|Pt
B.Pt|Cl2(P)|HC1(aq)|AgCl(s)|Ag(s)
C.Ag(s)|AgN03(aq)||HCI(aq)|AgC1(s)|Ag(s)
D.Ag(s)|AgCl(s)|HCl(aq)||AgN03(aq)|Ag
A.Pt,H2(g)∣HCl (aq)∣AgCl(s),Ag(s)
B.Zn (s)∣H2SO4(aq)∣Cu (s)
C.Pb(s),PbSO4(s)∣H2SO4(aq)∣PbSO4(s),PbO2(s)
D.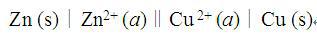
A.Zn|ZnCl2(aq)|Cl2(p)|Pt
B.Zn|ZnCl2(aq)|KCl(aq)|AgCl|Ag
C.Pt|H2(p1)|HCl(aq)|Cl2(p2)|Pt
D.Ag|AgCl(s)|KCl(aq)|Cl2(p)|Pt
(1) 2Al(s)+6HCl(aq)=Al2Cl6(aq)+3H2(g) ΔrHm(1)= -1007 kJ·mol-1 (2) H2(g)+Cl2(g)=2HCl(g) ΔrHm(2)= -184.8 kJ·mol-1 (3) HCl(g)+aq=HCl(aq) ΔrHm(3)= -72.73 kJ·mol-1 (4) Al2Cl6(s)+aq=Al2Cl6(aq) ΔrHm(4)= -645.5 kJ·mol-1 计算无水氯化铝的生成焓为________kJ·mol-1
往氨水中加少量下列物质时,NH3的解离度和溶液的:pH将发生怎样的变化? (1)NH4Cl(s) (2)NaOH(s) (3)HCl(aq) (4)H2O(1)
A.Ag|AgNO3(aq)||HCl(aq)|AgCl,Ag
B.Ag,AgCl|HCl(aq)|Cl2(p)(Pt)
C.(Pt)Cl2(p)|HCl(aq)||AgNO3(aq)|Ag
D.Ag,AgCl|HCl(aq)|AgCl,Ag


为了保护您的账号安全,请在“简答题”公众号进行验证,点击“官网服务”-“账号验证”后输入验证码“”完成验证,验证成功后方可继续查看答案!

 微信搜一搜
微信搜一搜
 简答题
简答题



 微信搜一搜
微信搜一搜
 简答题
简答题