题目内容
(请给出正确答案)
题目内容
(请给出正确答案)
由热力学可知,理想气体的焓只与温度有关,对组分i理想气体在某状态下的焓,可由摩尔定压热容计算According to thermodynamics, the enthalpy of ideal gas is only related to temperature. For component I, the enthalpy of ideal gas in a certain state can be calculated by molar heat capacity at constant pressure: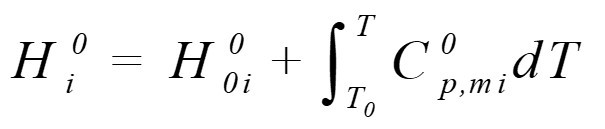 Cp,mi0—组分i理想气体摩尔定压热容Component I ideal gas molar heat capacity at constant pressure,kJ/(kmol?K)
Cp,mi0—组分i理想气体摩尔定压热容Component I ideal gas molar heat capacity at constant pressure,kJ/(kmol?K)

 如搜索结果不匹配,请
如搜索结果不匹配,请